Health
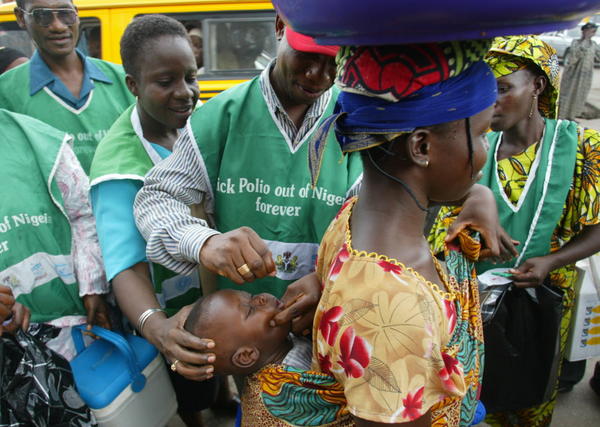
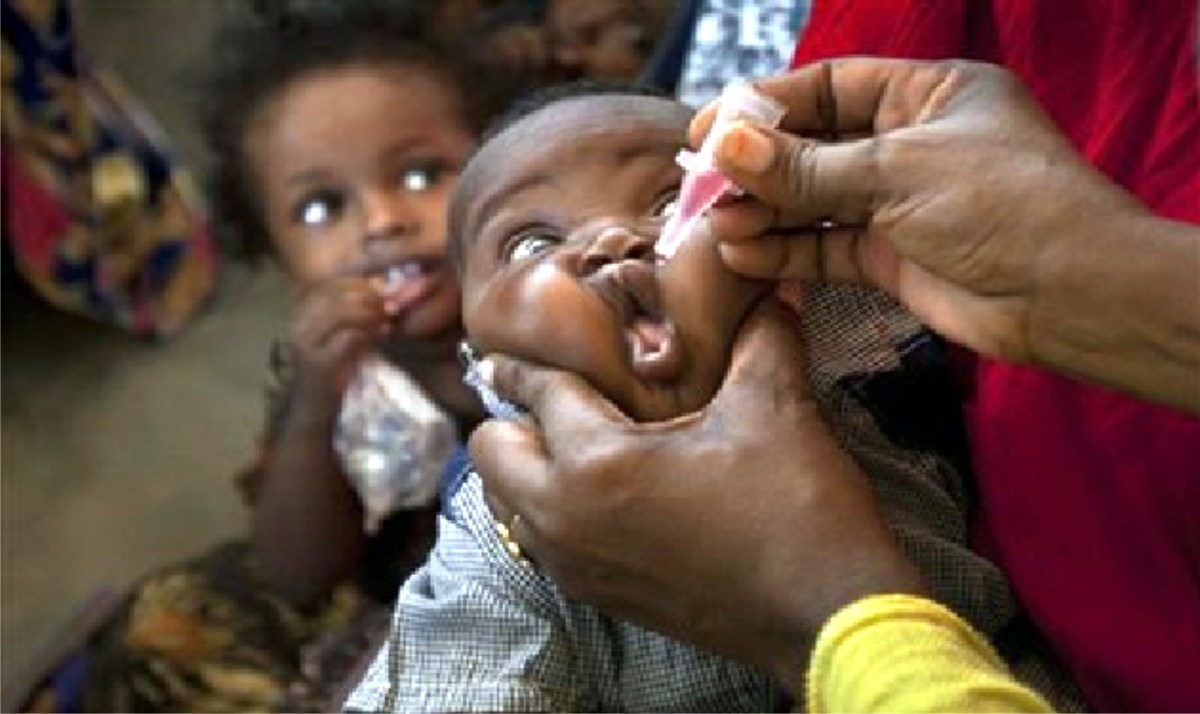
Kenya Kicks Off Polio Campaign
By Modupe Gbadeyanka
Kenya has kicked off its 2017 immunization effort with a polio campaign this week, targeting 2.9 million children aged five years and under.
The campaign, January 18-22, is targeting 15 counties that are that are considered on higher risk and vulnerable and which are mainly situated in remote, hard-to-reach and border areas.
The campaign also covers Nairobi County, the travel hub that brings the rest of the world into Kenya. The other counties include: Isiolo, Samburu, West Pokot, Turkana, Marsabit, Garissa, Tana River, Wajir, Lamu, Bungoma, Busia and Uasin Gishu, Trans Nzoia, Nairobi and Mandera.
The campaign or supplementary immunization activity (SIA) marks the country’s commitment to avert any possible polio outbreak given renewed threats of polio due to an outbreak in Borno State Nigeria last July and August.
Since then, countries in the Horn of Africa region have committed to initiate polio campaigns to ensure the region continues to be safe. The cases in Nigeria became evident after the conflict area associated with the Boko Haram insurgents in Borno State became more accessible.
Before then, Nigeria, Africa and the global polio eradication community had enjoyed some relief after Nigeria was removed from the list of polio endemic countries in 2015 after going without a case for a year.
Speaking at the national launch in Isiolo County, Director of Health Services Dr Jackson Kioko said there was need to vaccinate children in this campaign in order to improve the overall population immunity especially in areas where routine vaccination coverage was low.
He said the outbreak in Nigeria last August had put children in Kenya at risk. He gave assurance that the vaccine used for the campaign was safe and was exactly the same as that used for routine vaccination in all health facilities, public or private.
He said about three quarters of children under two (73 per cent) were fully immunized while the rest were either unimmunized or under-immunized.
WHO Country Representative Dr Rudi Eggers said that until poliovirus transmission was interrupted in all endemic countries, all countries including Kenya still remained at risk of importation of polio.
“This is particularly true of vulnerable countries (including some of Kenya’s neighbours) with weak public health and immunization services and travel or trade links to endemic countries.”
In addition, he said due to the low polio vaccination coverage rates found in some counties, there were many children left vulnerable to the disease even in Kenya.
He called on the need for a comprehensive approach in which all eligible children (0-2 years) are reached with all the life-saving routine immunization vaccines (BCG, Measles, Pentavalent, Rota, Yellow Fever, IPV and others), regardless of where they are born, who they are or where they live. In remarks read by Dr Iheoma Onuekwusi, EPI lead WHO Kenya, Dr Eggers said evidence had shown that one out of every five children were missed by routine immunization services in Kenya as a whole and many more in remote, and hard to reach areas in Kenya. He said for effective disease control and eradication, there was need to strengthen and address gaps in routine immunization services and the surveillance system.
“To secure and maintain a polio free world, we must reach every last child with the polio vaccine through Routine Immunization services and during immunization campaigns.”
The launch was attended by among others Isiolo County governor, Godana Doyo who said vaccination was a sure way of protecting the child’s health and future.
Polio ambassador in Isiolo County Mohamed Abdulahi said: “If my parents had enabled my vaccination, I would not be dependent on other people for mobility (“Wazazi wangu wangenipatia chanjo ya polio singekuwa nasukumwa na gari ya kusukumwa”).
National polio ambassador Senator Harold Kipchumba urged parents and neighbours to get their children vaccinated, adding that once infected by the virus, the disability could not be reversed.
“All you need are two drops to protect the child from polio,” he said. Dr Eggers also said no efforts were being spared to interrupt transmission of the Wild Polio Virus (WPV) in the endemic countries. These are Pakistan, Afghanistan and Nigeria. In 2016, 35 polio cases caused by wild type virus were detected from these three countries he added, in a speech read by Dr Iheoma Onuekwusi, EPI lead at WHO Kenya. He said the wild Poliovirus type 2 (WPV2) had been eradicated completely in 1999 while type 3 was virtually on the verge of eradication since it has not been detected anywhere in the world since November 2012. “The remaining strain, Type 1 WPV, could be eradicated with more effort on our part”, he added.
How real is the threat of polio?
The threat is real and has to be taken seriously, given the pattern in the last few outbreaks which emerged from the West African region. The last outbreak in 2013 also originating in Nigeria found its way into Kenya through Somalia. This was followed by numerous campaigns to ensure that every child was reached and through efforts to strengthen routine immunization and surveillance.
The last Horn of Africa Technical Advisory Group (TAG) meeting in September called on countries to avert any re-importation of polio. Dr Jean-Marc Olive, the chair of the TAG, said that given the
population movement pathways from West Africa to the region, the immunity surveillance gaps and declining routine immunization and previous history in Horn of Africa countries, the region was
vulnerable to an importation of the virus.
“After polio virus was identified in Borno state in Nigeria, we have to ask ourselves if there could be a Borno-like situation in our countries in the Horn-of-Africa, where we are missing transmission for a long period,” he said. Each country needed to find its ‘weakest point’ that they would focus on, he added. The virus in Nigeria is believed to have circulated without being detected for about five years and had possibly been exported to neighboring countries. Borno State is an insecure area whose access had been hampered by the Boko Haram insurgents and surveillance severely limited with close to half of settlements inaccessible.
Health
Resident Doctors Suspend Proposed Indefinite Strike
By Adedapo Adesanya
The Nigerian Association of Resident Doctors (NARD) has suspended its planned indefinite strike following the federal government’s reversal of the implementation of the reviewed Professional Allowance Table (PAT) and renewed assurances on outstanding payments.
The decision was announced in a communiqué issued at the end of an emergency National Executive Council (NEC) meeting held virtually on Saturday.
NARD had earlier resolved to embark on a total and indefinite strike over the government’s suspension of the reviewed allowance structure and other unresolved welfare concerns affecting resident doctors nationwide.
However, the association said it reconsidered its position after reviewing the outcomes of high-level engagements with key government officials and health-sector stakeholders.
According to the communiqué signed by NARD President, Dr Mohammad Usman Suleiman; Secretary-General, Dr Shuaibu Ibrahim; and Publicity and Social Secretary, Dr Abdulmajid Yahya Ibrahim, the Federal Government has now reversed its earlier decision on the allowance table.
“The NEC observed that the earlier decision to halt the implementation of the reviewed Professional Allowance Table (PAT) has been reversed, with implementation expected to reflect in the April salary and beyond,” the statement read.
The association also noted the government’s renewed commitment to settling outstanding promotion and salary arrears owed to resident doctors in affected institutions.
In addition, NARD said initial approval had been secured for the 2026 Medical Residency Training Fund (MRTF), with assurances that the disbursement process would be concluded.
“The NEC observed that the Budget Office has indicated its readiness to commence the process for the payment of the outstanding nineteen months’ arrears of the Professional Allowance,” the communiqué added.
Despite the progress, the doctors expressed concern about the continued delay in paying house officers’ salaries and called for urgent action to address the issue.
Following its deliberations, the NEC demanded the sustained implementation of the reviewed allowance structure, the prompt payment of all outstanding arrears, and the expedited disbursement of the residency training fund.
It also called for the immediate commencement of the process to clear the 19-month arrears and the convening of an urgent stakeholders’ meeting to resolve delays affecting house officers’ salaries.
“In light of the above developments, the NEC resolves to suspend the proposed total, indefinite, and comprehensive strike action, with a review of progress to be undertaken at the May Ordinary General Meeting (OGM) in Kano,” the statement said.
NARD expressed appreciation to President Bola Tinubu, Vice President Kashim Shettima, and several ministers, government agencies, and stakeholders for their interventions in resolving the dispute.
Health
Over 1.5 million Nigerian Children Living With Sickle Cell Disease—Report
By Modupe Gbadeyanka
More than 1.5 million children under the age of 15 are living with sickle cell disease in Nigeria, a new international study published in The Lancet Child & Adolescent Health, one of the world’s leading medical journals, has revealed.
In the report made available to Business Post, it was disclosed that Nigeria carries the highest burden of disease globally, far exceeding other high-burden countries such as the Democratic Republic of the Congo and Ethiopia.
The findings highlight both the scale of the challenge in Nigeria and the opportunity for the country to lead Africa in tackling one of the most preventable causes of childhood illness and death.
The study shows that nearly nine million children across sub-Saharan Africa are living with sickle cell disease in 2023, including around 1.17 million infants and 2.75 million children under five, who face the highest risk of early death without treatment.
Sickle cell disease is an inherited blood disorder present at birth. With early diagnosis and access to simple, low-cost interventions such as newborn screening, penicillin prophylaxis, routine vaccinations, malaria prevention, and hydroxyurea, most complications and deaths can be prevented.
However, in Nigeria, access to these essential services remains limited. Many children are only diagnosed after severe and avoidable complications, while others are never diagnosed at all, contributing to high levels of preventable illness and early childhood deaths.
The researchers emphasise that strengthening Nigeria’s health system response will be critical. This includes expanding newborn screening programmes, improving access to essential medicines, and integrating sickle cell care into primary healthcare services.
They called for urgent and coordinated action across government, health institutions, and development partners, including expanding newborn screening programmes, improving access to essential medicines and vaccines, and embedding sickle cell care within primary healthcare services.
The researchers, led by Professor Davies Adeloye, Professor of Public Health at Teesside University, United Kingdom, and Director of the International Society of Global Health (ISoGH), also called for increased domestic investment, supported by international partnerships, as well as stronger data systems to improve surveillance and guide policy decisions.
They concluded that even modest improvements in early-life screening and treatment in high-burden countries like Nigeria could transform child survival and significantly reduce preventable deaths.
“Nigeria now stands at the centre of the global sickle cell crisis. With over 1.5 million children affected, the scale is enormous, but so is the opportunity to act. We already know what works. Newborn screening and early treatment are effective, affordable, and can be delivered through existing health systems.
“If Nigeria prioritises sickle cell disease within its national health agenda and integrates care into routine maternal and child health services, we could save hundreds of thousands of young lives and significantly reduce avoidable deaths.” Professor Adeloye noted.
It was learned that the study analysed data from 40 studies across 22 African countries to produce the most comprehensive country-level estimates of childhood sickle cell disease to date.
Health
Helical Secures $10m Funding Package for Expansion

By Dipo Olowookere
A $10 million capital has been raised by Helical to support expansion across more top-20 pharma programmes and growth of its deployed science engineering team.
The firm will also use the money to build the compounding evidence layer that improves performance across diseases, as its mission is to make every scientist able to test hypotheses at the speed of inference and to turn in-silico discovery into a reliable engine for R&D throughput.
The funding package was from redalpine, Gradient, BoxGroup, Frst and notable angels, including Aidan Gomez (CEO Cohere), Clement Delangue (CEO HuggingFace) and Mario Goetze (pro soccer player).
Helical has a product known as the virtual AI lab for pharma, an application layer that turns biological foundation models into decision-ready, reproducible in-silico discovery workflows.
The platform has two product surfaces — the Virtual Lab for biologists and translational scientists, and the Model Factory for ML engineers and data scientists — built on the same data, the same models, and the same results.
By putting both sides in the same system, Helical closes the gap between computational predictions and biological decision-making, so teams that traditionally worked in silos can collaborate on the same evidence.
Helical was founded in early 2024. It was created by three school friends who took different paths to the same problem.
Rick Schneider built tech at Amazon and later helped the German enterprise Celonis scale in France and Japan. Maxime Allard led data science teams at IBM before pursuing a PhD focused on reinforcement learning and robotics. Mathieu Klop became a cardiologist and genomics researcher.
When bio foundation models emerged, the trio saw the chance to build the missing application layer that would let pharma teams move from model experimentation to reproducible, production discovery.
“The models alone don’t discover drugs. The system does. Pharma teams need a system that turns foundation models into workflows scientists can run, validate, and defend.
“We built Helical to make in-silico science reproducible at pharma scale, so teams can go from hypothesis to decision in days instead of months,” the co-founder of Helical, Mr Rick Schneider, said.
“We are at a unique point in time where biological foundation models and general language reasoning models are converging.
“We backed Helical because we strongly believe they have what it takes to build the pharma AI orchestration platform that will drive this transition from siloed AI models to integrated virtual AI labs,” the General Partner at redalpine, Mr Daniel Graf, stated.
-

 Feature/OPED6 years ago
Feature/OPED6 years agoDavos was Different this year
-
Travel/Tourism10 years ago
Lagos Seals Western Lodge Hotel In Ikorodu
-

 Showbiz3 years ago
Showbiz3 years agoEstranged Lover Releases Videos of Empress Njamah Bathing
-

 Banking8 years ago
Banking8 years agoSort Codes of GTBank Branches in Nigeria
-

 Economy3 years ago
Economy3 years agoSubsidy Removal: CNG at N130 Per Litre Cheaper Than Petrol—IPMAN
-

 Banking3 years ago
Banking3 years agoSort Codes of UBA Branches in Nigeria
-

 Banking3 years ago
Banking3 years agoFirst Bank Announces Planned Downtime
-

 Sports3 years ago
Sports3 years agoHighest Paid Nigerian Footballer – How Much Do Nigerian Footballers Earn