Health
COVAX to Facilitate Equal Access to COVID-19 Vaccine—WHO
By Adedapo Adesanya
The World Health Organisation (WHO) said the international community has established the COVAX Facility aimed at ensuring all countries have equal access to the coronavirus disease vaccines.
The head of the world health body, Mr Tedros Adhanom Ghebreyesus, said on Friday that there was an urgent need to address the financing gap to provide COVID-19 vaccines for everyone and everywhere.
In an appeal for stepped-up funding to support poorer countries, Mr Tedros said “this week, the United Kingdom began the rollout of a vaccine developed by pharmaceutical companies, Pfizer and BioNTech, and more nations are expected to follow suit very soon.”
Mr Tedros, speaking during his regular briefing from Geneva, Switzerland, further said: “To have safe and effective vaccines against a virus that was completely unknown to us only a year ago is an astounding scientific achievement.
“But an even greater achievement would be to ensure that all countries enjoy the benefits of science equitably.
“The international community has established a mechanism, known as the COVAX Facility, aimed at ensuring all countries will have equal access to any vaccines, once developed.”
Nearly 190 countries are participating, and the goal is to deliver two billion doses by the end of 2021.
Mr Tedros said there was an immediate funding gap of $4.3 billion to procure vaccines for the neediest countries.
“I urge donors to fill this gap quickly so that vaccines can be secured, lives can be saved and a truly global economic recovery is accelerated,” he stated.
The WHO chief also called on world leaders to translate political commitment for equitable vaccine access into action.
Meanwhile, the UN agency and its partners are helping countries to strengthen their supply chains in preparation for delivery.
Mr Tedros reported that nearly one billion doses of three vaccine candidates have already been secured, and further deals will be announced in the near future.
Evaluation of the first requests from countries eligible for assistance under the COVAX Facility is also underway.
WHO will soon be making its own determination as to whether some COVID-19 vaccines will be ready for rollout, a senior official said on Friday in response to a journalist’s question.
Several manufacturers have been submitting trial data to the WHO for emergency-use licensing.
Chief Scientist, Mr Soumya Swaminathan, said only those with Phase 3 clinical trial results would be considered.
“We started with the Pfizer dossier; we expect also to have the Moderna followed by the AstraZeneca dossiers examined in the next few weeks,” she said. “And we will be coming out with the decision whether it is receiving an emergency use license or not.”
Mrs Swaminathan added that WHO is working with the International Coalition for Medical Regulatory Agencies (ICMRA) “to speed up things further”. Several national regulators have also volunteered to assist with the assessments.
Her colleague, Mr Bruce Aylward, WHO Senior Adviser, explained that these processes were established to meet the goal of providing vaccines for all.
“We are indeed looking at these products though the WHO Emergency Use Listing Procedure,” he said.
“At the same time, we have an exceptional procedure in place where some products that are approved by what we call a stringent regulatory authority, can also be considered by the COVAX Facility, so there will be no barrier to the speed with which these products could potentially be used globally.”
Mrs Swaminathan recommended that countries will need to have national vaccination plans and related communications strategies in place.
It is important for authorities to explain the deployment process to citizens “because things are happening extremely fast and people are anxious for information”.
She said surveys indicate that most of the world’s population want a COVID-19 vaccine, “but at the same time, many do have questions concerning the process”
“Given that doses initially will be in limited supply, the public also needs to understand why priority will be given to frontline workers, the elderly and other at-risk groups.
“And the more open and transparent we can be, the more likely it is that people will have the trust and confidence and would not only want to take the vaccine but would also be patient and wait for their turn,” she said.
Health
Jacaranda Gets Funds to Expand Affordable Maternal Healthcare in Kenya

By Modupe Gbadeyanka
To expand affordable healthcare in Kenya, Swedfund has invested about $600,000 into Jacaranda Health Limited (Jacaranda Maternity) to support innovations in neonatal intensive care and strengthen Jacaranda’s ability to provide life-saving services to underserved populations.
Jacaranda Maternity provides high-quality maternal health care at more affordable pricing than typical private providers, focusing on women in Nairobi’s low- and middle-income communities.
The new funding will support the opening of new hospitals, upgrading of neonatal care, and improvements to existing facilities.
Maternal and newborn health outcomes in Kenya remain a challenge, with maternal mortality still high despite improvements in skilled birth attendance.
Public health facilities play a central role but face capacity constraints, while access to reliable, quality care varies across regions and income groups.
Private healthcare providers offering essential maternity services at accessible price points can complement public provision.
Jacaranda Maternity aims to expand its network to six hospitals to achieve financial sustainability while scaling its impact. The healthcare provider is a recognised leader in promoting women’s health, with 71 percent of its staff being women, and a track record of effective environmental and social management.
“This investment will help Jacaranda Maternity provide life-saving care to more women and families while furthering Swedfund’s mission to promote inclusive and sustainable healthcare,” a Senior Investment Manager at Swedfund, Audrey Obara, said.
Health
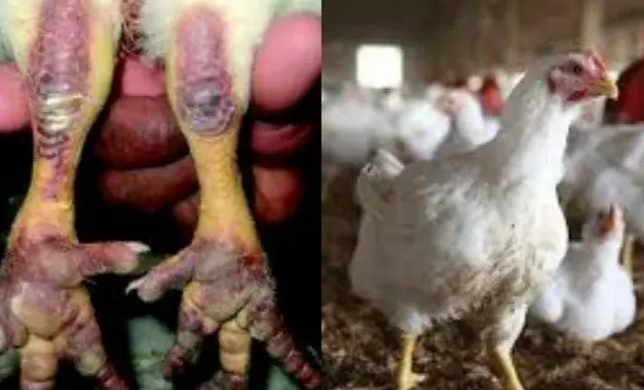
Nigeria Secures $350,000 FAO Support to Tackle Rising Bird Flu
By Adedapo Adesanya
Nigeria will get a $350,000 intervention from the Food and Agriculture Organisation of the United Nations (FAO) to support its response to the ongoing outbreak of Highly Pathogenic Avian Influenza (bird flu) and strengthen the country’s animal health systems.
An agreement was reached on Wednesday during a strategic meeting between the Minister of Livestock Development, Mr Idi Mukhtar Maiha, and the FAO Representative to Nigeria and the Economic Community of West African States, Mr Hussein Gadain, in Abuja.
The intervention, approved under FAO’s Technical Cooperation Programme, will support disease containment efforts in 11 affected states and enhance surveillance, coordination and response mechanisms to prevent further spread of the disease.
Speaking during the meeting, Maiha said effective disease control remains critical to improving livestock productivity and protecting the livelihoods of farmers across the country.
He explained that factors such as drought, scarcity of feed, interaction between livestock and wildlife, as well as cross-border movement of animals have contributed to the spread of diseases in some areas.
“We must continue to strengthen our animal health systems and build the capacity required to respond effectively to disease outbreaks. Our collaboration with FAO will help protect livestock assets, improve productivity and support the broader transformation of the sector,” the minister said.
Mr Gadain commended the federal government’s commitment to the development of the livestock sector and assured that FAO would continue to provide technical support to Nigeria.
He stressed the need to strengthen veterinary services at the state and community levels, improve early detection of diseases and promote biosecurity practices among livestock farmers.
The meeting also reviewed progress on the global campaign to eradicate Peste des Petits Ruminants, a highly contagious disease that affects sheep and goats.
To advance the initiative, the ministry plans to convene a national technical meeting involving veterinary institutions, researchers and practitioners to review Nigeria’s eradication strategy and address gaps in vaccine supply.
As part of preparations, the ministry will engage the National Veterinary Research Institute to assess its vaccine production capacity while exploring other options for vaccine procurement to meet national demand.
Both parties also agreed to accelerate Nigeria’s access to financing under the Pandemic Fund through the One Health approach in collaboration with the Nigeria Centre for Disease Control and the Federal Ministry of Health to strengthen preparedness and response to zoonotic diseases.
Plans are also underway for the Director-General of FAO to participate in the Antimicrobial Resistance Conference scheduled for June 2026 in Abuja, where President Bola Tinubu is expected to be recognised as the African Champion for the eradication of Peste des Petits Ruminants.
The meeting further agreed to inaugurate a Livestock Donor Working Group to coordinate development partner support and advance key initiatives, including the development of a national feed and fodder strategy aimed at improving productivity and sustainability in the livestock sector.
Health
Chimamanda: Euracare Raises Concerns Over MDCN Investigation Panel Process

By Aduragbemi Omiyale
A Lagos-based healthcare facility currently in the limelight, Euracare Multi-Specialist Hospital, has faulted the outcome of the investigation panel of the Medical and Dental Council of Nigeria (MDCN) on the death of a 21-month-old Nkanu Nnamdi Esege, son of a renowned author, Chimamanda Ngozi Adichie.
The toddler died some weeks ago after an alleged overdose of sedative propofol, with the family alleging medical negligence.
This week, the panel suspended the two doctors of Euracare, Dr Tosin Majekodunmi and Dr Titus Ogundare.
Reacting to the development in a statement, the hospital claimed it observed “a number of serious concerns that have arisen in the course of these proceedings.”
In the statement made available to Business Post, Euracare emphasised that it vouches for the “professionalism and integrity of our clinical team,” pointing out that “certain established processes and protocols have not been followed in the manner required” during the probe.
While it empathised “with the family of Master Nkanu Nnamdi Esege” over the unfortunate incident, the healthcare firm said there was a “serious breach” by the investigators that “cannot go unaddressed.”
It identified this breach as the disclosure of “matters covered by patient and institutional confidentiality” outside the appropriate channels.
Below is the full statement from Euracare;
Our attention has been drawn to widespread media reports concerning the interim suspension orders and other findings issued by the Medical and Dental Practitioners Investigation Panel against thirteen doctors, two of whom are our clinical staff members in connection with the ongoing proceedings relating to the death of Master Nkanu Nnamdi Esege. We remain fully committed to cooperating with all relevant regulatory and judicial authorities in the course of their inquiries.
We however wish to place on record our confidence in the professionalism and integrity of our clinical team. Dr. Tosin Majekodunmi and Dr. Titus Ogundare who are experienced professionals whose records of service to patients in Nigeria span many years. Both doctors have, in their respective careers, contributed meaningfully to the delivery of quality healthcare to Nigerian patients at a standard comparable to what is obtainable in the world’s leading medical facilities.
In the interest of transparency, since the commencement of this matter, we have conducted a thorough internal review of the clinical events in question, in line with our clinical governance standards and best practices. We have actively demonstrated our commitment to transparency and will continue to engage openly with all inquiries directed at us.
We are also compelled to draw attention to a number of serious concerns that have arisen in the course of these proceedings. It is our position that certain established processes and protocols have not been followed in the manner required. We have further noted, with deep concern, that matters covered by patient and institutional confidentiality appear to have been disclosed outside the appropriate channels, and we consider this a serious breach that cannot go unaddressed.
We wish to state that we stand by the principles of equality, fairness, and good governance. Every party in this matter, including our institution and our staff, is entitled to a process that is conducted with rigour, impartiality, and respect for the rules that govern it. We will be raising these concerns through the appropriate legal and regulatory channels.
We continue to empathize with the family of Master Nkanu Nnamdi Esege. The loss of a child is a grief without measure, and we carry that awareness in everything we say and do in relation to this matter.
-

 Feature/OPED6 years ago
Feature/OPED6 years agoDavos was Different this year
-
Travel/Tourism10 years ago
Lagos Seals Western Lodge Hotel In Ikorodu
-

 Showbiz3 years ago
Showbiz3 years agoEstranged Lover Releases Videos of Empress Njamah Bathing
-

 Banking8 years ago
Banking8 years agoSort Codes of GTBank Branches in Nigeria
-

 Economy3 years ago
Economy3 years agoSubsidy Removal: CNG at N130 Per Litre Cheaper Than Petrol—IPMAN
-

 Banking3 years ago
Banking3 years agoSort Codes of UBA Branches in Nigeria
-

 Banking3 years ago
Banking3 years agoFirst Bank Announces Planned Downtime
-

 Sports3 years ago
Sports3 years agoHighest Paid Nigerian Footballer – How Much Do Nigerian Footballers Earn

