Health
Accurate Multi-Panel Drug Test Cups For Professionals

In safety-sensitive workplaces, clinical settings, and staffing operations, reliable on-site drug screening is a practical first line of defense. Accurate multi-panel drug test cups for professionals combine speed with portability, allowing organizations to screen for multiple substances quickly while preserving chain-of-custody and sample integrity. This guide explains how these cups work, how to choose the right product for a professional setting, and how to manage administration, interpretation, and compliance to reduce legal risk and costly false positives.
What Multi-Panel Drug Test Cups Are And Who Should Use Them
Multi-panel drug test cups are self-contained immunoassay devices that screen a urine sample for multiple drug classes simultaneously, typically including amphetamines, cocaine metabolites, opioids, cannabinoids (THC), benzodiazepines, and others depending on the panel. They are designed for point-of-care use: a donor provides a urine specimen directly into the cup, and the integrated test strips produce rapid visual results within minutes.
Who should use them? Professionals that benefit most include:
- Occupational health and human resources teams conducting pre-employment, post-incident, or random testing programs.
- Substance use clinicians and treatment centers performing routine monitoring.
- Staffing firms and temp agencies that need quick screening before placement.
- Corrections and probation officers performing supervision checks.
- Employers in transportation, construction, healthcare, and manufacturing where safety is critical.
For organizations prioritizing both speed and defensibility, multi-panel cups offer a pragmatic balance: they provide immediate screening to inform next steps while still allowing for confirmatory laboratory testing when required.
How Multi-Panel Test Cups Work: Technology And Accuracy Factors
At their core, most multi-panel cups use lateral flow immunoassay technology. Antibodies embedded on test strips bind to drug metabolites in the specimen: a visible line forms (or disappears) according to the assay design, indicating a negative or presumptive positive.
Key accuracy factors to understand:
- Antibody specificity and cross-reactivity: High-quality assays use antibodies selected to minimize cross-reactivity with over-the-counter medications or endogenous compounds. Lower-grade tests may yield false positives when donors take legal medications that share similar metabolites.
- Cutoff concentrations: Each assay has a cutoff (measured in ng/mL) that determines whether a result is reported as positive. Most professional cups follow SAMHSA or DOT cutoffs for workplace testing: knowing these thresholds reduces misinterpretation.
- Temperature and matrix checks: Modern cups often integrate temperature strips and creatinine/oxidant checks to detect dilution or tampering. These integrity features improve the reliability of on-site results.
- Operator influence: Proper collection, timing, and result reading windows directly affect accuracy. Even the best cup can produce incorrect readings if the test is read too early or too late.
Real-world accuracy is often expressed in sensitivity (ability to detect positives) and specificity (ability to rule out negatives). Professional cups from reputable manufacturers typically report >95% agreement with laboratory immunoassays at the stated cutoffs, though confirmatory GC-MS or LC-MS/MS remains the gold standard for legal or employment consequences.
Choosing The Right Cup For Professional Settings
Selecting the appropriate multi-panel drug test cup requires more than picking a high panel count. It’s about matching features to use case, workflow, and legal requirements.
Key Selection Criteria
- Regulatory alignment: Choose cups that adhere to SAMHSA/DOT cutoffs if testing falls under federal guidelines.
- Built-in integrity checks: Temperature, adulteration, and creatinine tests help detect tampering or dilution at collection.
- Ease of use: Simple, unambiguous results and clear timing windows reduce operator error and training burden.
- Documentation options: Cups that help clear labeling, lot tracking, and photo documentation streamline chain-of-custody.
- Shelf life and storage needs: Longer shelf life and uncomplicated storage conditions simplify inventory management.
Panel Selection: Which Drugs To Include
Common professional panels are 5-, 8-, or 12-panel cups. Decisions should be risk-based:
- 5-panel: Standard workplace screens (AMP, COC, OPI, THC, BZO).
- 8–12 panel: Add methamphetamine, oxycodone, fentanyl, barbiturates, PCP, and others where clinical or workplace exposure warrants it.
Staffing and healthcare employers often opt for expanded panels that include fentanyl and synthetic opioids given their prevalence.
Sensitivity, Cutoffs, And False Positives
Higher sensitivity isn’t always better: it may detect clinically irrelevant low levels or passive exposure. Professional programs typically use established cutoffs to balance sensitivity and specificity. When a presumptive positive appears, organizations must have a policy for confirmatory testing rather than making employment decisions on a single cup result.
Adulteration Detection And Integrity Features
Buy cups with built-in tamper indicators: temperature strips (correct collection window), creatinine or pH checks, and oxidant detection. These features lower the likelihood of undetected sample manipulation and strengthen the defensibility of the screening process.
Best Practices For Collection And Administration
Accurate results start with consistent collection procedures and well-trained staff.
Chain Of Custody And Documentation
Maintain an unbroken chain of custody: donor identification, time-stamped collection, witness signatures, and secure transport for confirmatory samples. Use standardized forms and consider photo documentation or barcode systems that tie cups to donor records. This reduces disputes and protects both employer and donor.
Proper Sample Collection And Handling Steps
- Verify donor identity and inspect the collection area for prohibited items.
- Instruct the donor to provide an adequate volume into the cup: record temperature immediately.
- Observe, where policy and law permit, to prevent substitution.
- Seal and label samples promptly if they will be sent for confirmatory testing.
- Adhere to manufacturers’ timing for reading results: most cups specify a 5–10 minute window.
Training Staff And Reducing Human Error
Regular staff training on procedure, result interpretation, and documentation reduces mistakes. Provide quick-reference guides, role-play common scenarios, and audit collections periodically to ensure compliance.
Interpreting Results And When To Confirm
On-site cups deliver presumptive results, actionable only within a clear policy framework.
Reading Immunoassay Results Correctly
Most cups use a two-line format: a control line indicating the test is valid, and a test line indicating a negative or positive depending on the device. A faint test line usually indicates a negative result at or above the cutoff: no test line indicates a presumptive positive. Staff should follow the manufacturer’s visual guide and timing strictly. Photographing results can help document ambiguous cases.
Confirmatory Testing: When And How To Send Samples
Any presumptive positive that could have employment consequences should be sent to a certified laboratory for confirmatory testing using gas chromatography–mass spectrometry (GC-MS) or liquid chromatography–tandem mass spectrometry (LC-MS/MS). It’s best practice to split or retain an aliquot during collection for immediate confirmatory shipment if required. Establish relationships with accredited labs and clarify reporting timeframes, cutoff standards, and evidence handling procedures.
Legal, Regulatory, And Privacy Considerations
Testing programs operate in a regulated and privacy-sensitive environment: mishandling can result in litigation or regulatory penalties.
Compliance With Workplace Testing Regulations
Understand federal, state, and industry-specific regulations that apply to the organization. Transportation and DOT-regulated employees face stricter protocols, specific cutoffs, and certified collector requirements. Non-DOT employers still should align policies with best practices to maintain defensibility.
Recordkeeping, Privacy, And Liability Best Practices
Limit access to test results to authorized personnel, store records securely, and retain documentation according to legal retention requirements. Clear written policies that explain testing rationale, disciplinary procedures, and the appeals process help mitigate liability. Include provisions for reasonable accommodations and medical review officers (MROs) who assess legitimate medical explanations for positive results.
Storage, Shelf Life, And Vendor Reliability
Purchasing decisions affect accuracy and supply-chain resilience.
Storage Conditions, Expiry, And Inventory Management
Store cups per manufacturer recommendations, usually at controlled room temperature away from direct sunlight. Track lot numbers and expiration dates in inventory systems and rotate stock using FIFO principles. Expired tests can degrade antibody performance and increase error rates.
Evaluating Suppliers And Quality Assurance Practices
Work with suppliers that provide lot-level COAs (Certificates of Analysis), recall notifications, and prompt customer support. Vet vendors for ISO or other quality certifications, clear warranty policies, and responsiveness to post-sale technical questions. For agencies that manage testing programs for clients, documented supplier reliability reduces operational risk and preserves client trust.
Conclusion
Accurate multi-panel drug test cups for professionals are a practical tool when used within a rigorously designed program: choose products that match regulatory requirements, incorporate integrity checks, and come from reliable vendors. Combine those choices with standardized collection procedures, trained staff, and clear confirmatory pathways to reduce false positives and legal exposure.
For agencies and businesses that support client organizations, whether staffing firms or occupational health providers, investing in the right cups and operational controls pays off in defensible screening results and smoother downstream workflows. When testing has real consequences, a presumptive result from a quality cup should be the beginning of a controlled process, not the final word.
Health
Jacaranda Gets Funds to Expand Affordable Maternal Healthcare in Kenya

By Modupe Gbadeyanka
To expand affordable healthcare in Kenya, Swedfund has invested about $600,000 into Jacaranda Health Limited (Jacaranda Maternity) to support innovations in neonatal intensive care and strengthen Jacaranda’s ability to provide life-saving services to underserved populations.
Jacaranda Maternity provides high-quality maternal health care at more affordable pricing than typical private providers, focusing on women in Nairobi’s low- and middle-income communities.
The new funding will support the opening of new hospitals, upgrading of neonatal care, and improvements to existing facilities.
Maternal and newborn health outcomes in Kenya remain a challenge, with maternal mortality still high despite improvements in skilled birth attendance.
Public health facilities play a central role but face capacity constraints, while access to reliable, quality care varies across regions and income groups.
Private healthcare providers offering essential maternity services at accessible price points can complement public provision.
Jacaranda Maternity aims to expand its network to six hospitals to achieve financial sustainability while scaling its impact. The healthcare provider is a recognised leader in promoting women’s health, with 71 percent of its staff being women, and a track record of effective environmental and social management.
“This investment will help Jacaranda Maternity provide life-saving care to more women and families while furthering Swedfund’s mission to promote inclusive and sustainable healthcare,” a Senior Investment Manager at Swedfund, Audrey Obara, said.
Health
Nigeria Secures $350,000 FAO Support to Tackle Rising Bird Flu
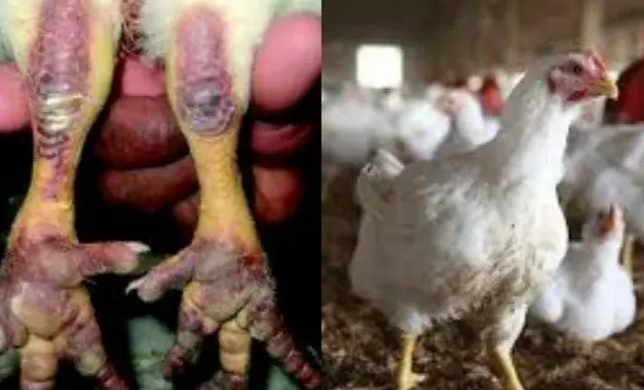
By Adedapo Adesanya
Nigeria will get a $350,000 intervention from the Food and Agriculture Organisation of the United Nations (FAO) to support its response to the ongoing outbreak of Highly Pathogenic Avian Influenza (bird flu) and strengthen the country’s animal health systems.
An agreement was reached on Wednesday during a strategic meeting between the Minister of Livestock Development, Mr Idi Mukhtar Maiha, and the FAO Representative to Nigeria and the Economic Community of West African States, Mr Hussein Gadain, in Abuja.
The intervention, approved under FAO’s Technical Cooperation Programme, will support disease containment efforts in 11 affected states and enhance surveillance, coordination and response mechanisms to prevent further spread of the disease.
Speaking during the meeting, Maiha said effective disease control remains critical to improving livestock productivity and protecting the livelihoods of farmers across the country.
He explained that factors such as drought, scarcity of feed, interaction between livestock and wildlife, as well as cross-border movement of animals have contributed to the spread of diseases in some areas.
“We must continue to strengthen our animal health systems and build the capacity required to respond effectively to disease outbreaks. Our collaboration with FAO will help protect livestock assets, improve productivity and support the broader transformation of the sector,” the minister said.
Mr Gadain commended the federal government’s commitment to the development of the livestock sector and assured that FAO would continue to provide technical support to Nigeria.
He stressed the need to strengthen veterinary services at the state and community levels, improve early detection of diseases and promote biosecurity practices among livestock farmers.
The meeting also reviewed progress on the global campaign to eradicate Peste des Petits Ruminants, a highly contagious disease that affects sheep and goats.
To advance the initiative, the ministry plans to convene a national technical meeting involving veterinary institutions, researchers and practitioners to review Nigeria’s eradication strategy and address gaps in vaccine supply.
As part of preparations, the ministry will engage the National Veterinary Research Institute to assess its vaccine production capacity while exploring other options for vaccine procurement to meet national demand.
Both parties also agreed to accelerate Nigeria’s access to financing under the Pandemic Fund through the One Health approach in collaboration with the Nigeria Centre for Disease Control and the Federal Ministry of Health to strengthen preparedness and response to zoonotic diseases.
Plans are also underway for the Director-General of FAO to participate in the Antimicrobial Resistance Conference scheduled for June 2026 in Abuja, where President Bola Tinubu is expected to be recognised as the African Champion for the eradication of Peste des Petits Ruminants.
The meeting further agreed to inaugurate a Livestock Donor Working Group to coordinate development partner support and advance key initiatives, including the development of a national feed and fodder strategy aimed at improving productivity and sustainability in the livestock sector.
Health
Chimamanda: Euracare Raises Concerns Over MDCN Investigation Panel Process

By Aduragbemi Omiyale
A Lagos-based healthcare facility currently in the limelight, Euracare Multi-Specialist Hospital, has faulted the outcome of the investigation panel of the Medical and Dental Council of Nigeria (MDCN) on the death of a 21-month-old Nkanu Nnamdi Esege, son of a renowned author, Chimamanda Ngozi Adichie.
The toddler died some weeks ago after an alleged overdose of sedative propofol, with the family alleging medical negligence.
This week, the panel suspended the two doctors of Euracare, Dr Tosin Majekodunmi and Dr Titus Ogundare.
Reacting to the development in a statement, the hospital claimed it observed “a number of serious concerns that have arisen in the course of these proceedings.”
In the statement made available to Business Post, Euracare emphasised that it vouches for the “professionalism and integrity of our clinical team,” pointing out that “certain established processes and protocols have not been followed in the manner required” during the probe.
While it empathised “with the family of Master Nkanu Nnamdi Esege” over the unfortunate incident, the healthcare firm said there was a “serious breach” by the investigators that “cannot go unaddressed.”
It identified this breach as the disclosure of “matters covered by patient and institutional confidentiality” outside the appropriate channels.
Below is the full statement from Euracare;
Our attention has been drawn to widespread media reports concerning the interim suspension orders and other findings issued by the Medical and Dental Practitioners Investigation Panel against thirteen doctors, two of whom are our clinical staff members in connection with the ongoing proceedings relating to the death of Master Nkanu Nnamdi Esege. We remain fully committed to cooperating with all relevant regulatory and judicial authorities in the course of their inquiries.
We however wish to place on record our confidence in the professionalism and integrity of our clinical team. Dr. Tosin Majekodunmi and Dr. Titus Ogundare who are experienced professionals whose records of service to patients in Nigeria span many years. Both doctors have, in their respective careers, contributed meaningfully to the delivery of quality healthcare to Nigerian patients at a standard comparable to what is obtainable in the world’s leading medical facilities.
In the interest of transparency, since the commencement of this matter, we have conducted a thorough internal review of the clinical events in question, in line with our clinical governance standards and best practices. We have actively demonstrated our commitment to transparency and will continue to engage openly with all inquiries directed at us.
We are also compelled to draw attention to a number of serious concerns that have arisen in the course of these proceedings. It is our position that certain established processes and protocols have not been followed in the manner required. We have further noted, with deep concern, that matters covered by patient and institutional confidentiality appear to have been disclosed outside the appropriate channels, and we consider this a serious breach that cannot go unaddressed.
We wish to state that we stand by the principles of equality, fairness, and good governance. Every party in this matter, including our institution and our staff, is entitled to a process that is conducted with rigour, impartiality, and respect for the rules that govern it. We will be raising these concerns through the appropriate legal and regulatory channels.
We continue to empathize with the family of Master Nkanu Nnamdi Esege. The loss of a child is a grief without measure, and we carry that awareness in everything we say and do in relation to this matter.
-

 Feature/OPED6 years ago
Feature/OPED6 years agoDavos was Different this year
-
Travel/Tourism10 years ago
Lagos Seals Western Lodge Hotel In Ikorodu
-

 Showbiz3 years ago
Showbiz3 years agoEstranged Lover Releases Videos of Empress Njamah Bathing
-

 Banking8 years ago
Banking8 years agoSort Codes of GTBank Branches in Nigeria
-

 Economy3 years ago
Economy3 years agoSubsidy Removal: CNG at N130 Per Litre Cheaper Than Petrol—IPMAN
-

 Banking3 years ago
Banking3 years agoSort Codes of UBA Branches in Nigeria
-

 Banking3 years ago
Banking3 years agoFirst Bank Announces Planned Downtime
-

 Sports3 years ago
Sports3 years agoHighest Paid Nigerian Footballer – How Much Do Nigerian Footballers Earn

