Health
Merck Unveils 2016 Hypertension Award Winners


By Dipo Olowookere
A leading science and technology company in healthcare, life science and performance materials, Merck, has announced the 2016 ‘Merck Hypertension Award’ winners to mark the World Hypertension Day.
Also, it has called for applications for the 2017 Hypertension Award, which is part of the Merck Capacity Advancement Program launched in April 2016 in partnership with African and Asian universities with the aim of building a platform of hypertension experts across the globe.
In Kampala, Uganda, Rasha Kelej, Senior Vice President and Chief Social Officer, Merck Healthcare emphasized: “Merck plays an important role in building diabetes and hypertension care capacity and improving access to quality and sustainable healthcare solutions in developing countries. Today marks an important day in the fight against hypertension as we mark the World Hypertension Day. In this context, we have announced ten Merck Hypertension Award 2016 winners.”
Merck invited medical students to apply for the “Merck Hypertension Award 2016” with the theme of “What the Healthy Heart needs.” Students across African and Asian medical universities were asked to submit a concept paper on how to improve hypertension awareness, early detection and prevention in their countries and how to encourage their society, scientific community, local authorities, media and relevant stakeholders to ‘think and act’ on hypertension every day.
The scientific committee received over 500 concept submission applications from universities in Africa and Asia and 10 winners were selected for the award. Winners from each university have been granted a one year postgraduate Preventive Cardiovascular Medicine diploma with the University of South Wales, United Kingdom.
“Merck Hypertension Award is another step in our long term commitment to support hypertension care strategy through working with local governments, academia and relevant stakeholders in building healthcare capacity with a focus on diabetes, hypertension and other non-communicable diseases in various countries in Asia-Pacific, Middle East, Africa and Latin America,” added Rasha Kelej when making the announcement of the winners during an event in Nairobi, Kenya.
Prof. Eligah Ogola, Vice President of Pan African Cardiology Society said: “We are happy to partner with Merck to implement their Capacity Advancement Program focusing on Hypertension in 2016. This combined diabetes and hypertension education program will contribute towards providing guidelines and clinical practice for prevention, diagnosis and management of these diseases and their complications. The objective of this initiative is to increase the level of knowledge for medical students who will work in the near future with patients to help them prevent, understand and control the diseases across the continent”.
Health
Ebola: NCAA Directs Enhanced Surveillance Across Nigerian Airports
By Adedapo Adesanya
The Nigeria Civil Aviation Authority (NCAA) has directed airlines, airport operators and other aviation stakeholders to intensify public health surveillance measures following the outbreak of Ebola Virus Disease (EVD) in parts of the Democratic Republic of Congo (DRC) and Uganda.
In a circular dated May 18, 2026, the NCAA said the directive became necessary due to growing concerns over the spread of the disease and the critical role of the aviation sector in preventing cross-border transmission.
The authority noted that although no confirmed case linked to the outbreak has been recorded in Nigeria, it is collaborating with the Federal Ministry of Health, the Nigeria Centre for Disease Control (NCDC), Port Health Services and international health organisations to closely monitor the situation.
According to the NCAA, health authorities in the DRC recently identified a cluster of severe illnesses among healthcare workers in the Bunia Health Zone in the northeastern part of the country.
Recall that Nigeria also said it would tighten surveillance after the World Health Organisation (WHO) declared a public health emergency after Ebola killed over 80 people in Congo and Uganda.
Laboratory investigations later confirmed the presence of the Bundibugyo virus, a strain of the Ebola virus family known to cause severe viral haemorrhagic disease in humans.
The authority stated that there is currently no licensed vaccine specifically approved for the Bundibugyo strain, while treatment remains largely supportive and symptom-based.
The NCAA listed symptoms associated with Ebola Virus Disease to include sudden fever, severe fatigue, persistent headache, vomiting, abdominal pain and bleeding manifestations such as nosebleeds or vomiting blood.
It stressed that early detection and immediate reporting remain critical to preventing the international spread of the disease.
As part of preventive measures, the authority said disease surveillance systems at airports have been strengthened, while contact tracing, case reporting mechanisms and border health screening procedures are also being reinforced.
The NCAA further directed pilots to notify Air Traffic Control of any suspected communicable disease cases onboard aircraft, in line with the Nigeria Civil Aviation Regulations.
It also instructed flight crew members to complete and submit Aircraft General Declaration forms for all suspected cases and mandated airlines to ensure passenger locator forms are completed and handed over to Port Health Services upon arrival.
The authority added that airlines must ensure aircraft are properly equipped with first aid kits, universal precaution kits and emergency medical kits.
It also urged operators to reinforce crew training on the identification and management of communicable diseases and ensure strict adherence to infection prevention and control guidelines.
The NCAA called on all aviation stakeholders to remain vigilant and comply fully with established public health protocols to safeguard passengers, crew members and the general public from potential health threats.
Health
Nigeria on High Alert as WHO Declares Ebola Emergency of International Concern
By Adedapo Adesanya
The Nigeria Centre for Disease Control and Prevention (NCDC) has said that although the country currently has no confirmed case of Ebola Virus Disease (EVD), it is, nevertheless, actively strengthening surveillance.
This comes as the World Health Organisation (WHO) declared the Ebola outbreak in Congo and Uganda a public health emergency of international concern, after 80 deaths were attributed to the disease.
The WHO, however, stopped short of declaring a pandemic, saying it did not meet the necessary criteria. The United Nations agency advised countries against closing borders or restricting trade.
Early symptoms include fever, muscle pain, fatigue, headache, and sore throat, and are followed by vomiting, diarrhoea, a rash, and bleeding.
In a statement by its Director General, Mr Jide Idris, on Sunday, the NCDC noted that it is also ramping up laboratory readiness, infection prevention, and public awareness efforts across the country.
He said the centre was closely monitoring the situation due to increasing regional movement across African countries and was working with relevant stakeholders, including the Port Health Services under the Federal Ministry of Health and Social Welfare, to strengthen preparedness within Nigeria’s public health system.
“NCDC is closely monitoring the situation and working with relevant stakeholders, including the Port Health Services, to ensure continued vigilance and preparedness within the public health system,” he stated.
The NCDC boss described Ebola virus disease as a severe viral illness transmitted through direct contact with the blood, bodily fluids, secretions, or contaminated materials of infected persons or animals.
He noted that the disease has an incubation period ranging from two to 21 days, while symptoms include fever, weakness, headache, muscle pain, sore throat, vomiting, diarrhoea, and, in severe cases, unexplained bleeding.
Recall that Nigeria gained international recognition for successfully containing an Ebola outbreak in 2014 after an infected traveller arrived in Lagos from Liberia.
Healthcare workers were advised to maintain a high index of suspicion for Ebola, especially in patients presenting symptoms compatible with the disease alongside relevant travel or exposure history.
Idris stressed the importance of strict adherence to infection prevention and control measures, including early identification and isolation of suspected cases, proper use of personal protective equipment, hand hygiene, and prompt reporting through established channels.
“NCDC will continue to monitor the situation closely and provide updates as necessary,” he added.
NCDC advised Nigerians to remain calm, maintain good hand hygiene, avoid misinformation, and report unusual illnesses promptly.
Health
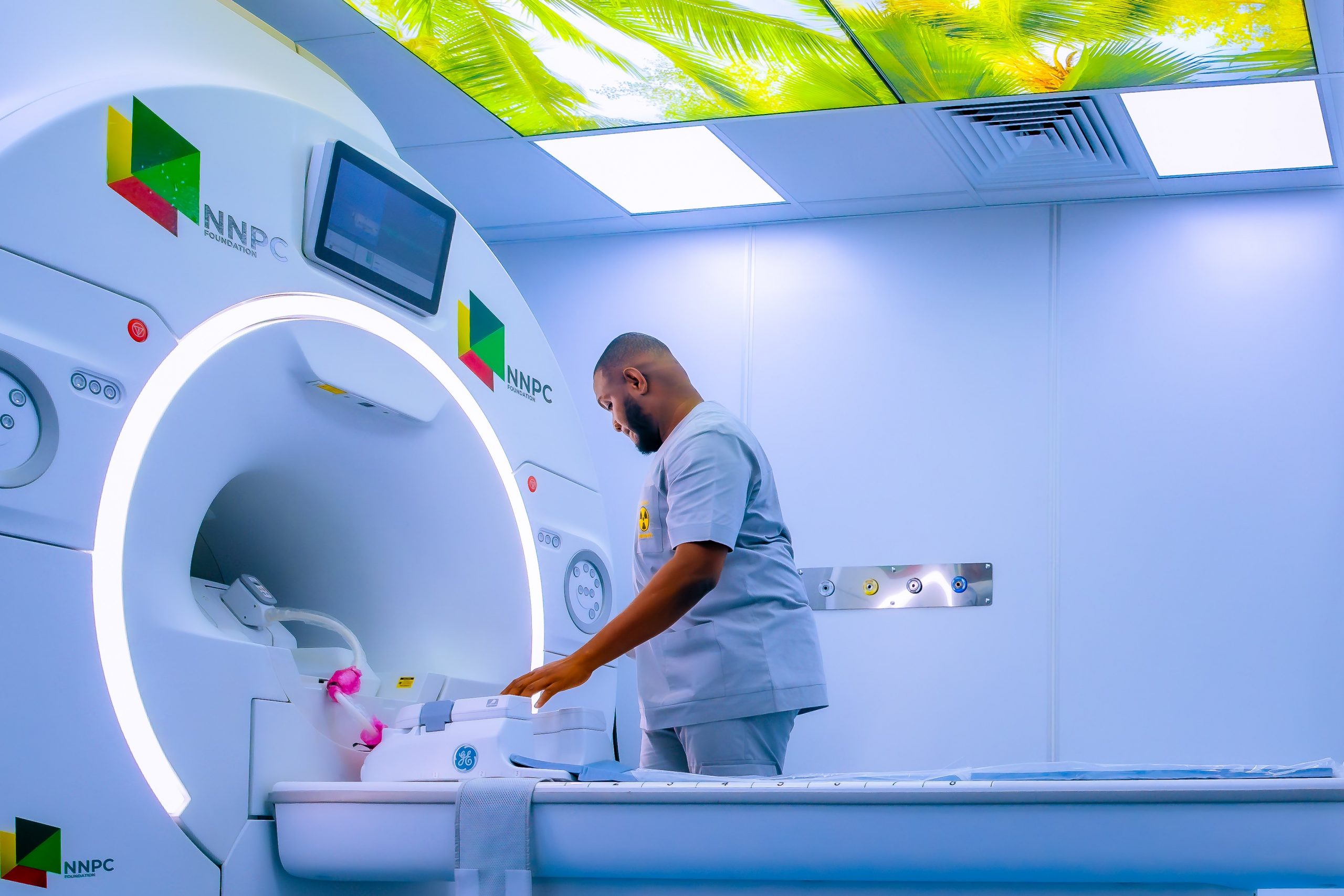
NNPC Donates MRI Machine, Others to Nnewi Teaching Hospital
By Modupe Gbadeyanka
A 1.5 Tesla Magnetic Resonance Imaging (MRI) machine has been donated to the Nnamdi Azikiwe University Teaching Hospital (NAUTH), Nnewi, Anambra State, by the corporate social responsibility arm of the Nigerian National Petroleum Company (NNPC) Limited, NNPC Foundation.
In a statement on Sunday by its Chief Corporate Communications Officer, Mr Andy Odeh, the state-owned oil organisation said the medical item was given to the healthcare institution as part of its commitment to improving healthcare access and strengthening medical infrastructure across Nigeria.
The MRI system is expected to significantly improve access to advanced diagnostic imaging services for millions of Nigerians across the South-East (Anambra, Enugu, Imo, Abia and Ebonyi States) as well as neighbouring Delta State.
The foundation also provided critical supporting infrastructure, including RF shielding systems, chillers, backup UPS systems, electrical installations, specialised imaging accessories, ventilation systems, CCTV and oxygen monitoring systems, intercom communication facilities, and other patient comfort technologies designed to ensure optimal operation of the facility.
Before now, patients requiring advanced MRI diagnostic services often faced prolonged waiting periods, exorbitant costs, and the burden of travelling long distances in search of functional imaging centres.
But it is believed that the intervention of the NNPC Foundation would provide succour to patients.
At the presentation of the items to the institution over the weekend, the chief executive of the NNPC, Mr Bashir Bayo Ojulari, represented by the Managing Director of NNPC Foundation, Mrs Emmanuella Arukwe, described the intervention as a strategic investment in healthcare access, diagnostic precision, and improved patient outcomes, noting that the facility aligns with the company’s commitment to building sustainable systems and impactful national institutions.
“The installation of the MRI in NAUTH exemplifies our commitment, as our intent is to build enduring institutions, sustainable systems and legacies. This intervention aligns with our conviction that access to quality healthcare underpins human dignity, longevity and economic productivity,” Mr Ojulari stated.
He described the company’s social investments as viable currencies that strengthen the relationship between the Company’s core mandate of providing and managing energy for Nigerians and meeting stakeholders’ expectations.
In his remarks, the Governor of Anambra State, Mr Charles Soludo, who was represented by the Commissioner for Health, Dr Afam Obidike, said the intervention would enhance safe and precise diagnosis and treatment for patients across the South-East region.
He also commended NNPC Foundation for donating the MRI facility to the state, noting that the intervention would significantly improve access to quality healthcare services for the people.
The Chief Medical Director of NAUTH, Prof Joseph Ugboaja, thanked the donor for the items, saying NNPC Foundation has demonstrated that corporate social responsibility is not just a policy statement but a lifeline for institutions like ours.
“For too long, patients in our catchment area have had to travel long distances to access this level of diagnostic precision, often at prohibitive costs. With this installation, we will eliminate that burden,” he enthused.
-

 Feature/OPED6 years ago
Feature/OPED6 years agoDavos was Different this year
-
Travel/Tourism10 years ago
Lagos Seals Western Lodge Hotel In Ikorodu
-

 Showbiz3 years ago
Showbiz3 years agoEstranged Lover Releases Videos of Empress Njamah Bathing
-

 Banking8 years ago
Banking8 years agoSort Codes of GTBank Branches in Nigeria
-

 Economy3 years ago
Economy3 years agoSubsidy Removal: CNG at N130 Per Litre Cheaper Than Petrol—IPMAN
-

 Banking3 years ago
Banking3 years agoSort Codes of UBA Branches in Nigeria
-

 Banking3 years ago
Banking3 years agoFirst Bank Announces Planned Downtime
-

 Sports3 years ago
Sports3 years agoHighest Paid Nigerian Footballer – How Much Do Nigerian Footballers Earn

