Health
Nigeria Excluded as Ghana, 11 Others Get Malaria Vaccines
By Adedapo Adesanya
Twelve countries across different regions of Africa, excluding Nigeria, are set to receive 18 million doses of the first-ever malaria vaccine over the next two years.
The rollout of the RTS,S/AS01 vaccine will be piloted in Ghana, Kenya, and Malawi under the Malaria Vaccine Implementation Programme countries.
Allocations were also made for new introductions in Benin, Burkina Faso, Burundi, Cameroon, the Democratic Republic of the Congo, Liberia, Niger, Sierra Leone and Uganda. This will see the introduction of the vaccine into their routine immunization programmes for the first time.
This is a critical step forward in the fight against one of the leading causes of death on the continent: Malaria. Malaria remains one of Africa’s deadliest diseases, killing nearly half a million children under the age of 5, and accounting for approximately 95 per cent of global malaria cases and 96 per cent of deaths in 2021.
The allocations have been determined through the application of the principles outlined in the Framework for allocation of limited malaria vaccine supply that prioritizes those doses to areas of highest need, where the risk of malaria illness and death among children is highest.
Since 2019, Ghana, Kenya and Malawi have been delivering the malaria vaccine through the Malaria Vaccine Implementation Programme (MVIP), coordinated by WHO and funded by Gavi, the Vaccine Alliance, the Global Fund to Fight AIDS, Tuberculosis and Malaria, and Unitaid.
The RTS,S/AS01 vaccine has been administered to more than 1.7 million children in Ghana, Kenya and Malawi since 2019 and has been shown to be safe and effective, resulting in both a substantial reduction in severe malaria and a fall in child deaths. At least 28 African countries have expressed interest in receiving the malaria vaccine.
This allocation round makes use of the supply of vaccine doses available to Gavi, Vaccine Alliance via the United Nations Children’s Fund (UNICEF). The first doses of the vaccine are expected to arrive in countries during the last quarter of 2023, with countries starting to roll them out by early 2024.
“This vaccine has the potential to be very impactful in the fight against malaria, and when broadly deployed alongside other interventions, it can prevent tens of thousands of future deaths every year,” said Thabani Maphosa, Managing Director of Country Programmes Delivery at Gavi, the Vaccine Alliance.
“While we work with manufacturers to help ramp up supply, we need to make sure the doses that we do have are used as effectively as possible, which means applying all the learnings from our pilot programmes as we broaden out to a new total of 12 countries.”
“Nearly every minute, a child under 5 years old dies of malaria,” said UNICEF Associate Director of Immunization Ephrem T Lemango. “For a long time, these deaths have been preventable and treatable; but the roll-out of this vaccine will give children, especially in Africa, an even better chance at surviving. As supply increases, we hope even more children can benefit from this life-saving advancement.”
“The malaria vaccine is a breakthrough to improve child health and child survival, and families and communities rightly want this vaccine for their children. This first allocation of malaria vaccine doses is prioritised for children at highest risk of dying of malaria,” said Dr Kate O’Brien, WHO Director of Immunization, Vaccines and Biologicals.
“The high demand for the vaccine and the strong reach of childhood immunisation will increase equity in access to malaria prevention and save many young lives. We will work tirelessly to increase supply until all children at risk have access,” she added.
Annual global demand for malaria vaccines is estimated at 40–60 million doses by 2026 alone, growing to 80–100 million doses each year by 2030. In addition to the RTS,S/AS01 vaccine, developed and produced by GSK, and in the future supplied by Bharat Biotech, it is expected that a second vaccine, R21/Matrix-M, developed by Oxford University and manufactured by Serum Institute of India (SII), could also be prequalified by WHO soon. Gavi has recently outlined its roadmap to support increasing supply to meet demand.
Health
NARD Suspends Indefinite Strike, Gives FG Fresh Two-Week Ultimatum

By Adedapo Adesanya
The Nigerian Association of Resident Doctors (NARD) has suspended its planned nationwide indefinite strike, granting the federal government a two-week ultimatum to address lingering welfare issues affecting resident doctors across the country.
The decision was taken after an emergency meeting of the association’s National Executive Council on Tuesday, where members reviewed assurances from government representatives and resolved to give dialogue another chance.
NARD said the suspension was informed by “progress made” in negotiations, particularly commitments on the prompt payment of salary arrears, hazard allowances, and steps toward resolving issues surrounding the Medical Residency Training Fund.
The association did not declare a full resolution of the dispute. It noted that the government had shown “renewed willingness” to address the concerns that triggered the strike threat.
The association noted that while these engagements signalled a willingness by the government to resolve the dispute, several critical issues remain outstanding, particularly the delayed payment of promotion arrears, salary arrears, the 2026 Medical Residency Training Fund (MRTF), and the backlog of 19 months’ professional allowance arrears owed to resident doctors.
It also expressed concern over the Federal Government’s decision to halt the implementation of the reviewed PAT, which had earlier triggered widespread dissatisfaction among its members and raised fears of disruption to healthcare services nationwide.
Despite these unresolved issues, NARD said it opted to suspend the strike as a demonstration of goodwill and commitment to ongoing dialogue, while giving the government a two-week window to take concrete, measurable and verifiable steps to meet its demands.
The association insisted on the immediate reversal of the decision affecting the PAT, payment of all outstanding arrears, prompt disbursement of the MRTF, and full settlement of the accumulated professional allowance backlog.
It warned that it would reconvene at the expiration of the ultimatum to assess the level of compliance and determine its next course of action, adding that failure by the government to meet its demands within the stipulated timeframe would result in the resumption of the suspended strike without further notice.
NARD also called on its members nationwide to remain calm, united and resolute, while urging the Federal Government to act swiftly to prevent a potential crisis in the health sector.
The association further appreciated the interventions of the Vice President and other stakeholders, expressing hope that their involvement would lead to the timely resolution of the dispute and help sustain healthcare delivery across the country.
Health
Jacaranda Gets Funds to Expand Affordable Maternal Healthcare in Kenya

By Modupe Gbadeyanka
To expand affordable healthcare in Kenya, Swedfund has invested about $600,000 into Jacaranda Health Limited (Jacaranda Maternity) to support innovations in neonatal intensive care and strengthen Jacaranda’s ability to provide life-saving services to underserved populations.
Jacaranda Maternity provides high-quality maternal health care at more affordable pricing than typical private providers, focusing on women in Nairobi’s low- and middle-income communities.
The new funding will support the opening of new hospitals, upgrading of neonatal care, and improvements to existing facilities.
Maternal and newborn health outcomes in Kenya remain a challenge, with maternal mortality still high despite improvements in skilled birth attendance.
Public health facilities play a central role but face capacity constraints, while access to reliable, quality care varies across regions and income groups.
Private healthcare providers offering essential maternity services at accessible price points can complement public provision.
Jacaranda Maternity aims to expand its network to six hospitals to achieve financial sustainability while scaling its impact. The healthcare provider is a recognised leader in promoting women’s health, with 71 percent of its staff being women, and a track record of effective environmental and social management.
“This investment will help Jacaranda Maternity provide life-saving care to more women and families while furthering Swedfund’s mission to promote inclusive and sustainable healthcare,” a Senior Investment Manager at Swedfund, Audrey Obara, said.
Health
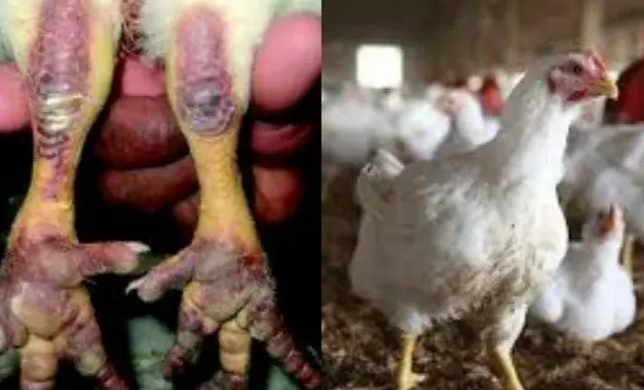
Nigeria Secures $350,000 FAO Support to Tackle Rising Bird Flu
By Adedapo Adesanya
Nigeria will get a $350,000 intervention from the Food and Agriculture Organisation of the United Nations (FAO) to support its response to the ongoing outbreak of Highly Pathogenic Avian Influenza (bird flu) and strengthen the country’s animal health systems.
An agreement was reached on Wednesday during a strategic meeting between the Minister of Livestock Development, Mr Idi Mukhtar Maiha, and the FAO Representative to Nigeria and the Economic Community of West African States, Mr Hussein Gadain, in Abuja.
The intervention, approved under FAO’s Technical Cooperation Programme, will support disease containment efforts in 11 affected states and enhance surveillance, coordination and response mechanisms to prevent further spread of the disease.
Speaking during the meeting, Maiha said effective disease control remains critical to improving livestock productivity and protecting the livelihoods of farmers across the country.
He explained that factors such as drought, scarcity of feed, interaction between livestock and wildlife, as well as cross-border movement of animals have contributed to the spread of diseases in some areas.
“We must continue to strengthen our animal health systems and build the capacity required to respond effectively to disease outbreaks. Our collaboration with FAO will help protect livestock assets, improve productivity and support the broader transformation of the sector,” the minister said.
Mr Gadain commended the federal government’s commitment to the development of the livestock sector and assured that FAO would continue to provide technical support to Nigeria.
He stressed the need to strengthen veterinary services at the state and community levels, improve early detection of diseases and promote biosecurity practices among livestock farmers.
The meeting also reviewed progress on the global campaign to eradicate Peste des Petits Ruminants, a highly contagious disease that affects sheep and goats.
To advance the initiative, the ministry plans to convene a national technical meeting involving veterinary institutions, researchers and practitioners to review Nigeria’s eradication strategy and address gaps in vaccine supply.
As part of preparations, the ministry will engage the National Veterinary Research Institute to assess its vaccine production capacity while exploring other options for vaccine procurement to meet national demand.
Both parties also agreed to accelerate Nigeria’s access to financing under the Pandemic Fund through the One Health approach in collaboration with the Nigeria Centre for Disease Control and the Federal Ministry of Health to strengthen preparedness and response to zoonotic diseases.
Plans are also underway for the Director-General of FAO to participate in the Antimicrobial Resistance Conference scheduled for June 2026 in Abuja, where President Bola Tinubu is expected to be recognised as the African Champion for the eradication of Peste des Petits Ruminants.
The meeting further agreed to inaugurate a Livestock Donor Working Group to coordinate development partner support and advance key initiatives, including the development of a national feed and fodder strategy aimed at improving productivity and sustainability in the livestock sector.
-

 Feature/OPED6 years ago
Feature/OPED6 years agoDavos was Different this year
-
Travel/Tourism10 years ago
Lagos Seals Western Lodge Hotel In Ikorodu
-

 Showbiz3 years ago
Showbiz3 years agoEstranged Lover Releases Videos of Empress Njamah Bathing
-

 Banking8 years ago
Banking8 years agoSort Codes of GTBank Branches in Nigeria
-

 Economy3 years ago
Economy3 years agoSubsidy Removal: CNG at N130 Per Litre Cheaper Than Petrol—IPMAN
-

 Banking3 years ago
Banking3 years agoSort Codes of UBA Branches in Nigeria
-

 Banking3 years ago
Banking3 years agoFirst Bank Announces Planned Downtime
-

 Sports3 years ago
Sports3 years agoHighest Paid Nigerian Footballer – How Much Do Nigerian Footballers Earn